Pre-exposure prophylaxis (PrEP) for HIV are medications that reduce an individual’s chances of contracting HIV. The CDC estimates that 1.2 million people are at risk of HIV infection and may benefit from a PrEP prescription.
We analyzed health insurance claims for 55 million Americans with employer-sponsored insurance in order to examine utilization and spending on these drugs between 2016 and 2020.
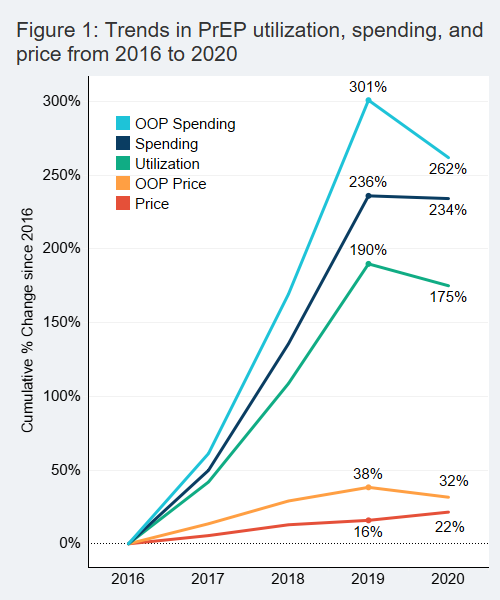
Between 2016 and 2019 the use of preexposure prophylaxis for HIV (PrEP) almost tripled for people with health insurance obtained through their job (Figure 1). Utilization dropped slightly from 2019 to 2020, likely due to the COVID-19 pandemic. Unlike some other medication, PrEP prescriptions require regular lab testing that must be performed in-person, which may have been an obstacle for some patients during the pandemic.
The price for a 30-day supply of PrEP increased steadily over the five years, growing 22%. The average point of sale price for a month’s supply of PrEP increased from $1,450 in 2016 to $1,761 in 2020 (Note: cost estimates do not include manufacturer rebates or coupons). Only branded versions of the two medications prescribed as PrEP, Truvada and Descovy, were available prior to 2021. Both Truvada and Descovy are manufactured by Gilead. Generic versions of Truvada became available in January 2021.
Patient out-of-pocket (OOP) price for PrEP increased 38% in the pre-pandemic period, from $94 for a month’s supply in 2016 to $130 in 2019.
PrEP Drug Availability Timeline
Several policy and regulatory changes have likely impacted prescribing patterns, utilization and spending on PrEP since it was first approved by the FDA in 2012.
- July 2012: Truvada approved by FDA for use as PrEP
- April 2016: Descovy came to market, although not yet approved for on-label use as PrEP
- June 2019: US Preventive Services Task Force rated PrEP as an effective preventive service for people at risk of HIV infection
- October 2019: Descovy was approved by FDA for use as PrEP (it could be prescribed as PrEP off-label in prior years)
- January 2021: Generic versions of Truvada became available
- January 2021: PrEP must be covered by insurers as a preventative with no patient-cost sharing under the Affordable Care Act
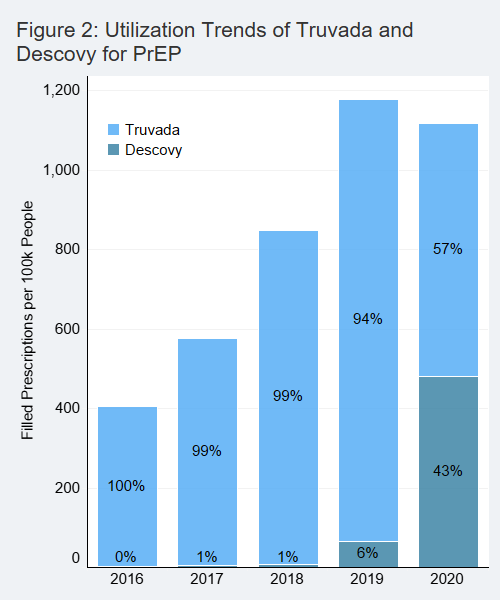
During this study period, 2016 through 2020, there were two medications approved for use as HIV pre-exposure prophylaxis: Truvada and Descovy. Descovy prescriptions saw a notable rise since the drug was approved for HIV PrEP in late 2019 (Figure 2). Over 40% of prescriptions for PrEP were for Descovy in 2020. Truvada and Descovy had similar point-of-sale prices.
Although utilization of PrEP increased substantially since 2016, these medications are still likely underutilized. Legal challenges to PrEP’s status as a no-cost preventative service could impact the affordability and availability of these medications in the future.
Limitations
These are trends for people with commercial health insurance obtained through their employer and may not reflect trends in other patient populations – Medicaid, Medicare, individual market, or uninsured. Claims data does include manufacturer rebates or patient copay coupons or discount programs, which may be commonly used for Truvada and Descovy.
People with medical claims with a listed diagnosis (ICD-10 codes B20, B97.35, R75, or Z21) were excluded from the analysis as medications used for PrEP may also be used as part of the treatment for HIV. Some people with HIV may not have had medical claims with an HIV diagnosis code listed and may not have been exclude from the analysis.
